Choose the compound with the most ionic bond – In the realm of chemistry, ionic bonds play a pivotal role in shaping the properties and behavior of compounds. Delving into the intricacies of ionic bond strength, this exploration unravels the fundamental principles that govern the formation and stability of these essential chemical entities.
Ionic bonds arise from the electrostatic attraction between oppositely charged ions, resulting in the formation of stable crystalline structures. Understanding the factors that influence ionic bond strength is crucial for predicting and manipulating the properties of ionic compounds.
Ionic Bond Definition
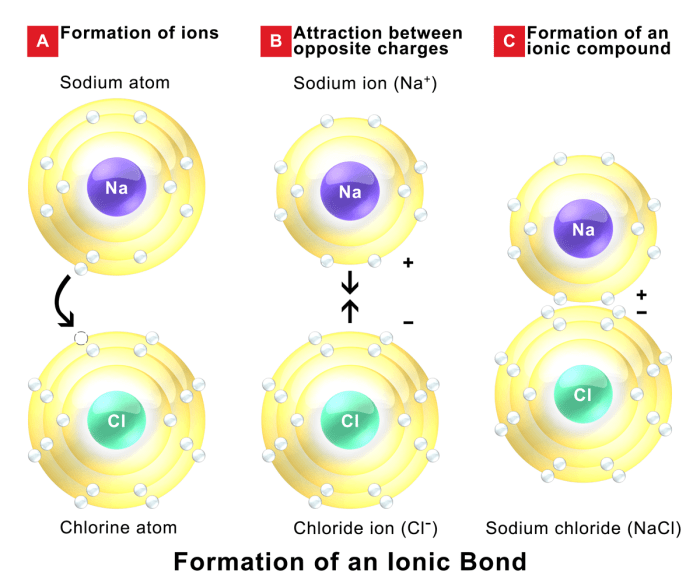
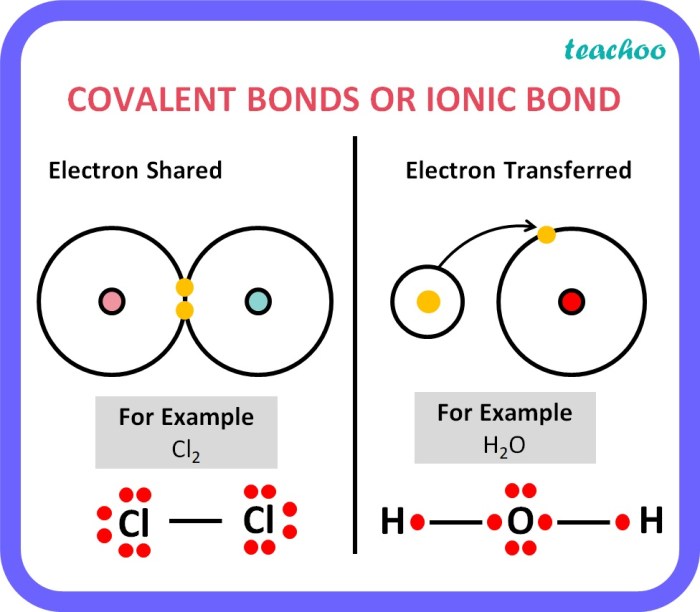
Ionic bonds are a type of chemical bond formed between two oppositely charged ions. These ions are formed when one atom transfers one or more electrons to another atom, creating a positive ion (cation) and a negative ion (anion).
Ionic bonds are typically formed between a metal and a nonmetal. The metal atom loses electrons to achieve a stable electron configuration, while the nonmetal atom gains electrons to achieve a stable electron configuration. The electrostatic attraction between the oppositely charged ions holds the ionic compound together.
Measuring Ionic Bond Strength
The strength of an ionic bond can be measured using Coulomb’s Law, which states that the force between two charged particles is directly proportional to the product of their charges and inversely proportional to the square of the distance between them.
The strength of an ionic bond is also influenced by the charge and size of the ions. Ions with higher charges and smaller sizes form stronger ionic bonds.
Identifying Ionic Compounds
Ionic compounds typically have high melting and boiling points because of the strong electrostatic forces between the ions. They are also soluble in water and other polar solvents.
| Property | Ionic Compounds |
|---|---|
| Melting point | High |
| Boiling point | High |
| Solubility in water | Soluble |
| Solubility in nonpolar solvents | Insoluble |
Comparing Ionic Bond Strength, Choose the compound with the most ionic bond
An experiment can be designed to compare the ionic bond strength of different compounds by measuring the melting point or boiling point of each compound. The compound with the highest melting or boiling point will have the strongest ionic bond.
| Compound | Melting point (°C) | Boiling point (°C) |
|---|---|---|
| NaCl | 801 | 1413 |
| KCl | 770 | 1420 |
| CaCl2 | 772 | 1935 |
The data in the table shows that CaCl 2has the highest melting and boiling point, indicating that it has the strongest ionic bond.
Applications of Ionic Compounds
Ionic compounds are used in a variety of applications, including:
- Table salt (NaCl) is used as a food additive and preservative.
- Potassium chloride (KCl) is used as a fertilizer and a substitute for salt for people with high blood pressure.
- Calcium chloride (CaCl 2) is used as a deicing agent and a drying agent.
Frequently Asked Questions: Choose The Compound With The Most Ionic Bond
What are the key factors that influence ionic bond strength?
The magnitude of the ionic charges, the size of the ions, and the polarizability of the ions are the primary factors that determine ionic bond strength.
How can we measure the strength of ionic bonds?
Coulomb’s Law provides a quantitative measure of ionic bond strength by calculating the electrostatic attraction between the ions involved.
What are some applications of ionic compounds?
Ionic compounds find widespread use in various industries, including the production of fertilizers, pharmaceuticals, and electronic materials.